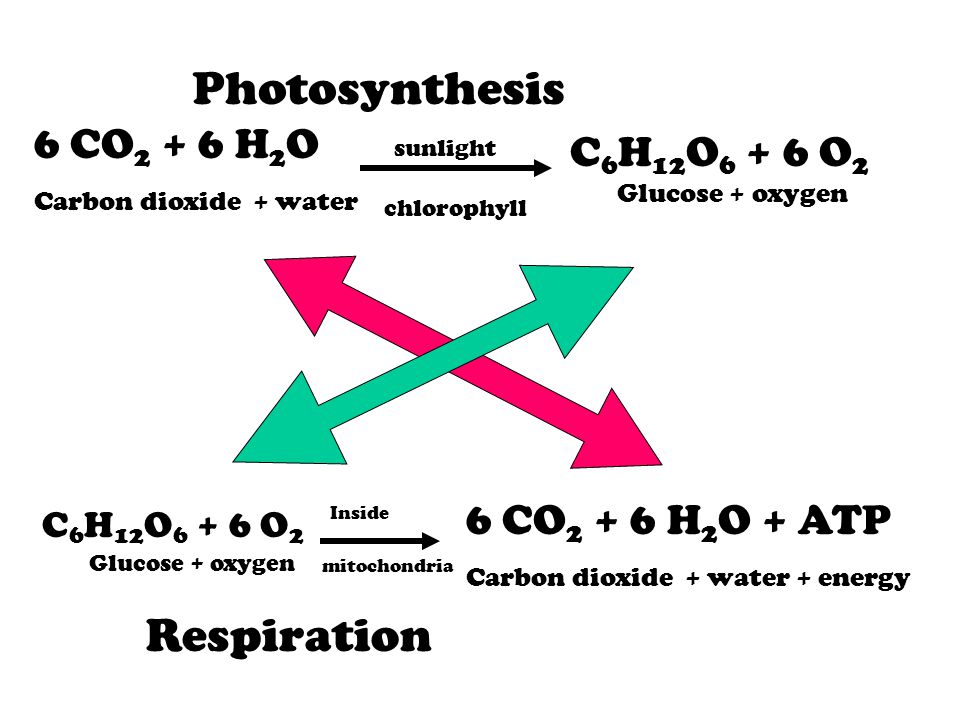
Photosynthesis chemical reaction (Remember… conservation of matter and energy) Light + 6 CO2 + 6 H2O C6H12O6 + 6 O2 + Heat **Remember to look for the. - ppt download

The structure diagrams of (H2O)6⁻ cluster isomers. The red and white... | Download Scientific Diagram
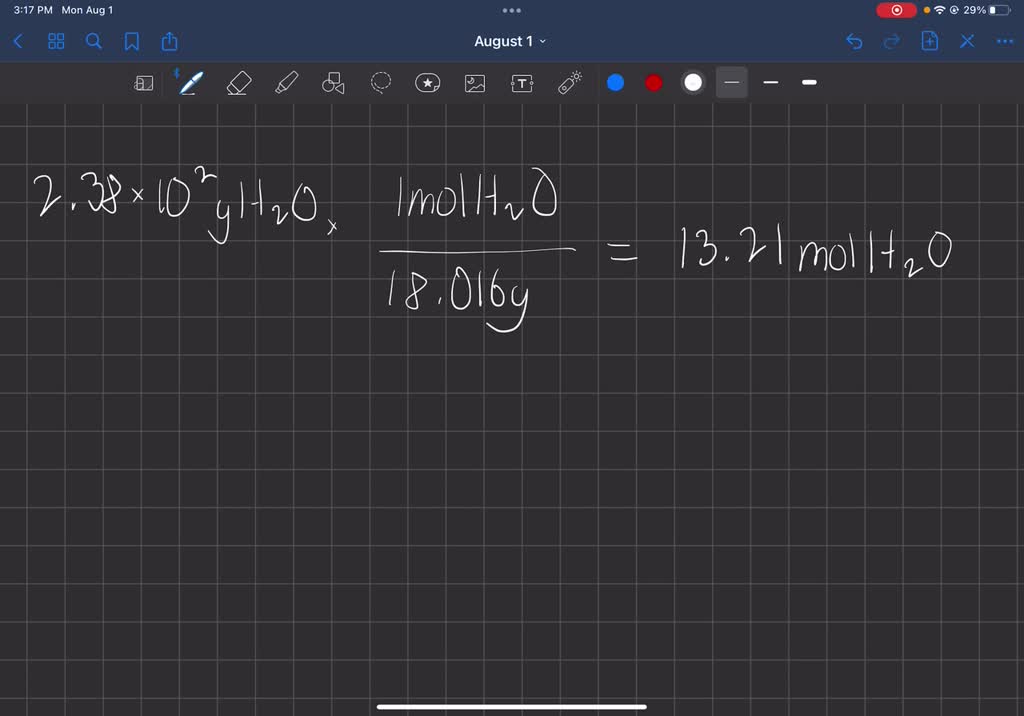
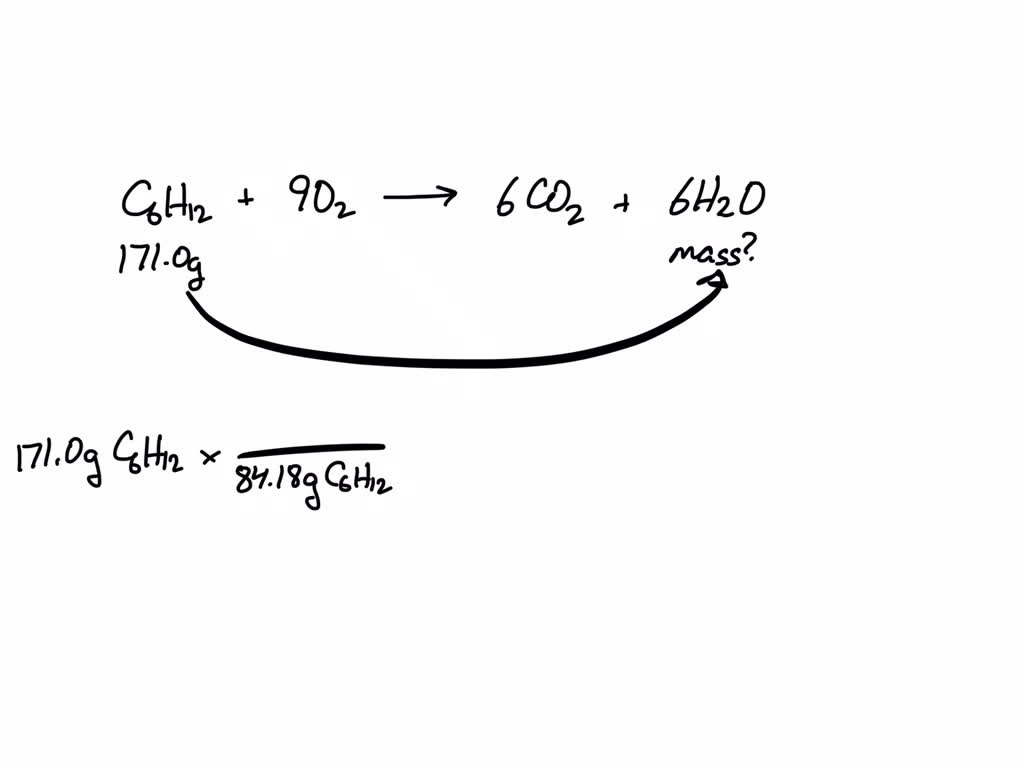
SOLVED: Consider this reaction: 6 CO2 + 6 H2O + light C6H12O6 + 6 O2If there were 2.38 x 102 g of H2O, 18.6 moles of CO2, and plenty of light, what would be the theoretical yield of the reaction?
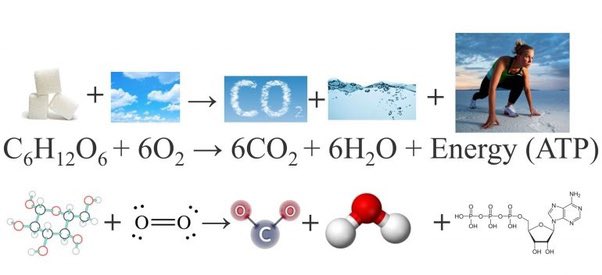
عبدالرحمن العتيبي on Twitter: "A simple example of a combustion reaction is the burning of methane: CH4(g) + 2 O2(g) → CO2(g) + 2 H2O(g)" / Twitter
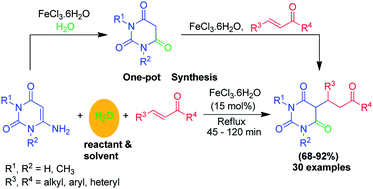
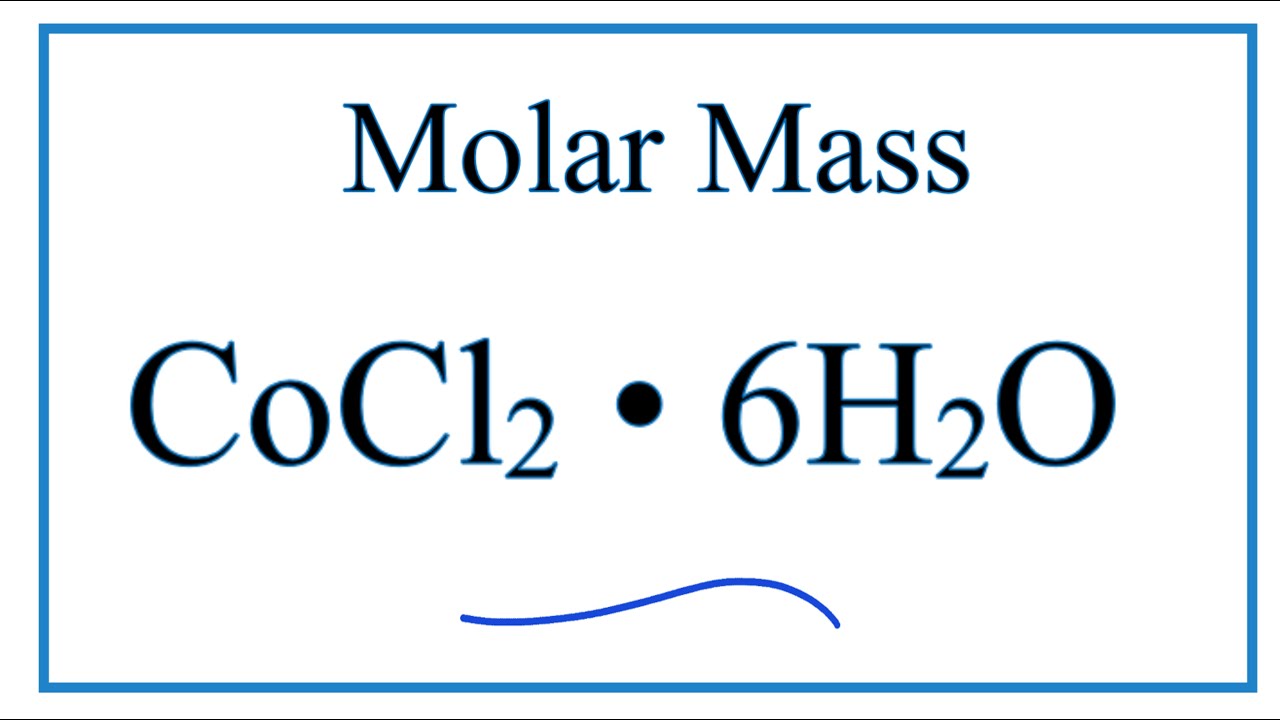
FeCl3·6H2O catalyzed aqueous media domino synthesis of 5-monoalkylbarbiturates: water as both reactant and solvent - RSC Advances (RSC Publishing)
![Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/jacs.8b11389/asset/images/jacs.8b11389.social.jpeg_v03)
Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society

Use the balanced equation to answer each question. 2 C2H6(g) + 5 O2(g) delta 4 CO(g) + 6 H2O(g) How - Brainly.com

The structure diagrams of (H2O)6⁻ cluster isomers. The red and white... | Download Scientific Diagram

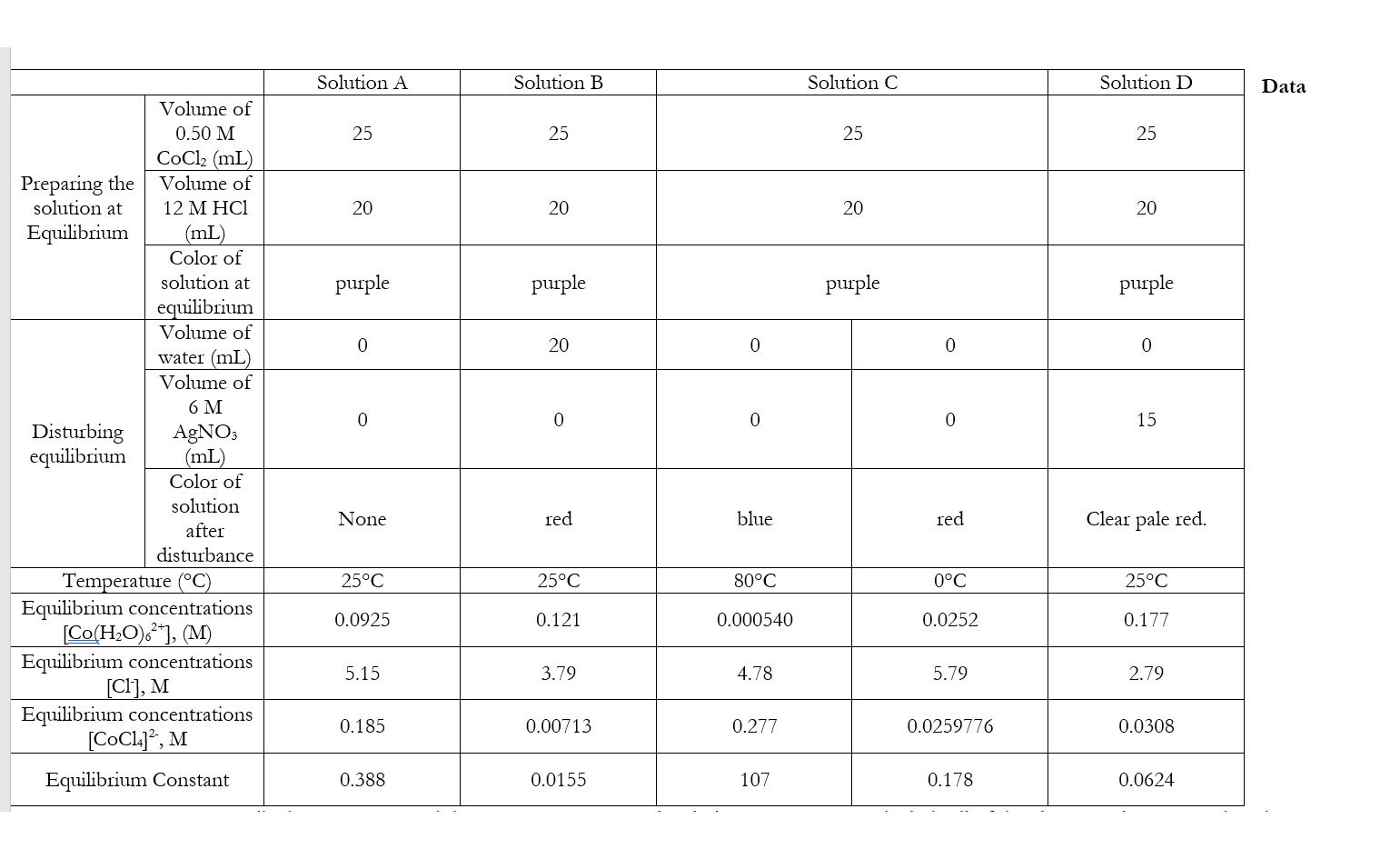
For the reaction: Co(H2O)6^{2+} + 4Cl^- \leftrightharpoons CoCl4^{2-} + 6H2O After adding 12M HCl, what would be the roles of H^+ ions and Cl^- ions? | Homework.Study.com
![Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation](https://pubs.acs.org/cms/10.1021/ct501143c/asset/images/medium/ct-2014-01143c_0017.gif)
Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation

Consider this reaction: 6 CO2 + 6 H2O + light equation C6H12O6 + 6 O2 If there were 2.38 x 102 g of - Brainly.in