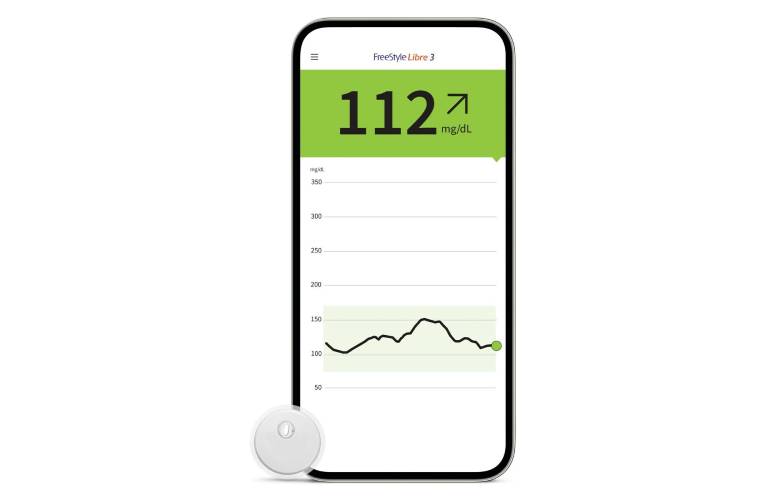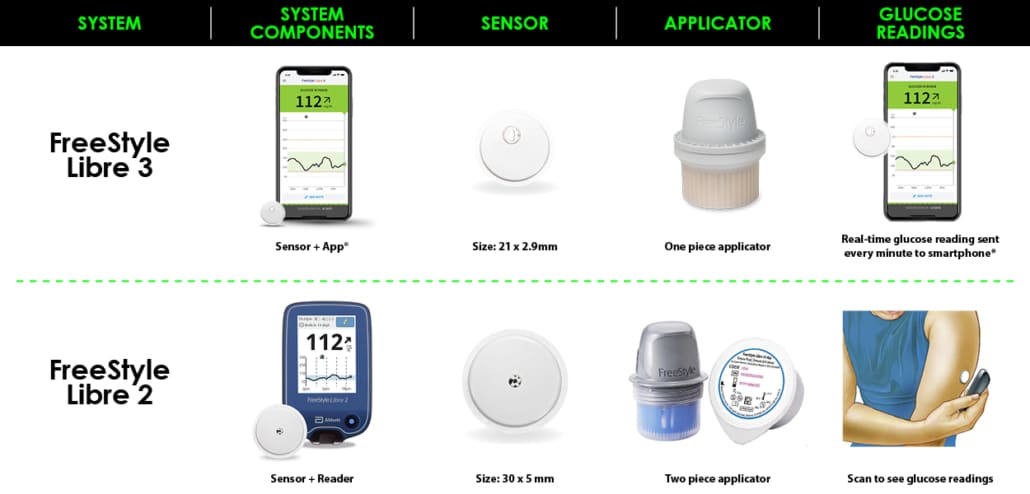
Abbott® Freestyle Libre 3 Sensor - Continuous Glucose Monitoring Device (CGM) - Advanced Diabetes Supply®

Amazon.com: Style Libre Free Sensor Cover | Hard, Durable | All Purpose USE from Sports to Shower| | FSL SENSOR COVER : Health & Household


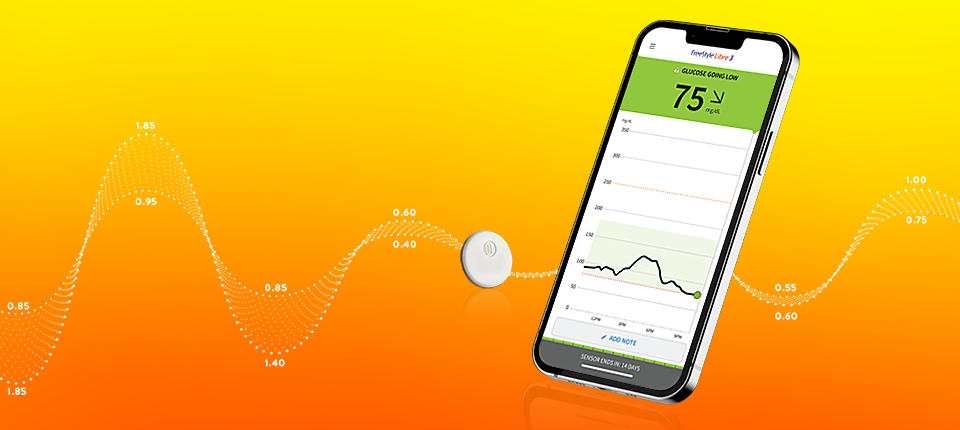
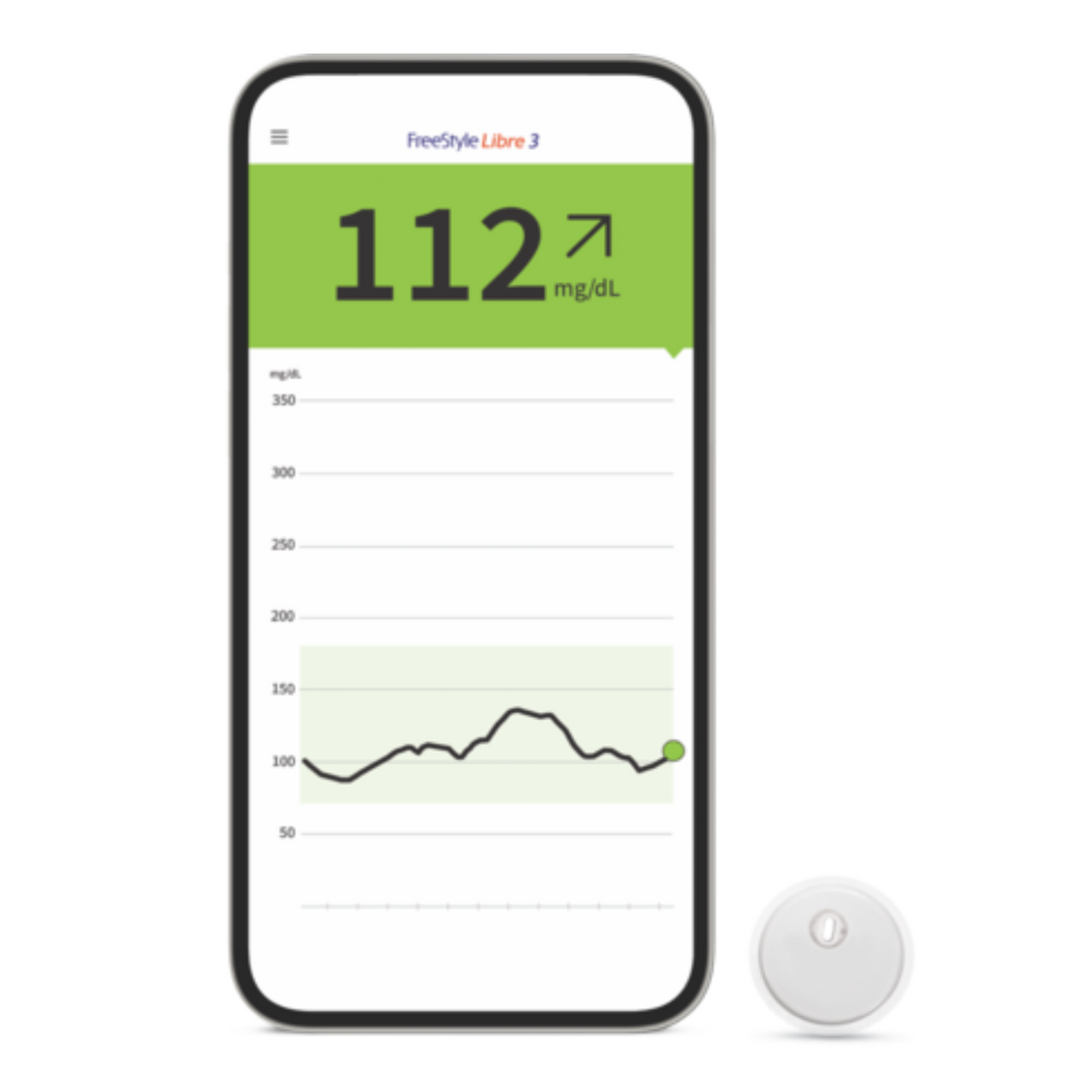
.jpg)