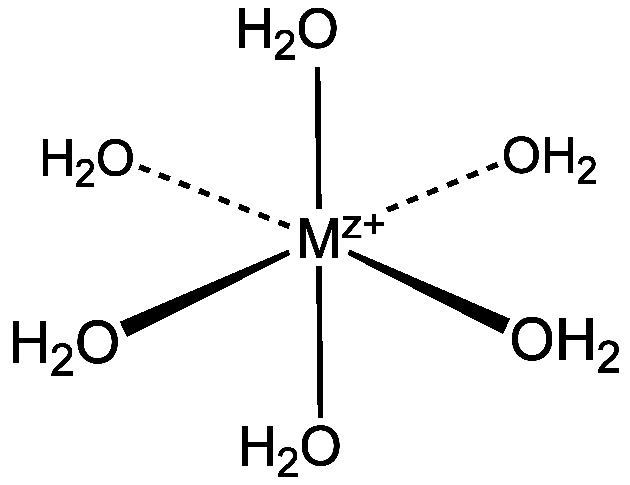
![Electronic Structure of 3d [M(H2O)6]3+ Ions from ScIII to FeIII: A Quantum Mechanical Study Based on DFT Computations and Natural Bond Orbital Analyses | Inorganic Chemistry Electronic Structure of 3d [M(H2O)6]3+ Ions from ScIII to FeIII: A Quantum Mechanical Study Based on DFT Computations and Natural Bond Orbital Analyses | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/ic001258t/asset/images/medium/ic001258tf00003.gif)
Electronic Structure of 3d [M(H2O)6]3+ Ions from ScIII to FeIII: A Quantum Mechanical Study Based on DFT Computations and Natural Bond Orbital Analyses | Inorganic Chemistry

Formaldehyde solution, 37 wt. % in H2O, concentration de 13.32 M | 50-00-0 | Reference standards | Alsachim

SOLVED: A mixture of 0.469 M H2O , 0.461 M Cl2O , and 0.664 M HClO are enclosed in a vessel at 25°C. H2O(g)+Cl2O(g)↽−−⇀2HOCl(g) ?c=0.0900 at 25°C Calculate the equilibrium concentrations of
![SOLVED: Calculate the equilibrium concentrations of H2O, Cl2, HCl, and O2 at 298 K if the initial concentrations are [H2O]=0.030M and [Cl2]=0.105M. The equilibrium constant Kc for the reaction H2O(g)+Cl2(g)⇌2HCl(g)+O2(g) is 8.96×10−9 SOLVED: Calculate the equilibrium concentrations of H2O, Cl2, HCl, and O2 at 298 K if the initial concentrations are [H2O]=0.030M and [Cl2]=0.105M. The equilibrium constant Kc for the reaction H2O(g)+Cl2(g)⇌2HCl(g)+O2(g) is 8.96×10−9](https://cdn.numerade.com/ask_previews/e62a205e-eaa0-4664-a57e-86d5123b2ca6_large.jpg)
SOLVED: Calculate the equilibrium concentrations of H2O, Cl2, HCl, and O2 at 298 K if the initial concentrations are [H2O]=0.030M and [Cl2]=0.105M. The equilibrium constant Kc for the reaction H2O(g)+Cl2(g)⇌2HCl(g)+O2(g) is 8.96×10−9

What is the height of H2O column (in m), thatcould be maintained by normal atmosphericpressure (= 1 - Brainly.in